Induced Pluripotent Stem Cells
- 4 days ago
- 3 min read
Induced pluripotent stem cells (iPSCs) represent one of the most transformative advances in modern biology. It was first reported by Shinya Yamanaka and colleagues in 2006. These iPSCs are adult (somatic) cells that have been genetically reprogrammed back into a pluripotent state-meaning they regain the ability to self-renew indefinitely and differentiate into virtually any cell type of the body.
This discovery fundamentally changed the landscape of stem cell research. By bypassing the ethical and practical limitations associated with embryonic stem cells (ESCs), iPSCs opened new avenues for disease modeling, regenerative medicine. , drug discovery, and personalized therapies.
What are Induced Pluripotent Stem Cells?
iPSCs are generated by reprogramming differentiated somatic cells- such as skin fibroblasts or blood cells through the forced expression of a small set of transcription factors. These factors reset the cell's epigenetic and transcriptional landscape, returnign it to pluripotent, embryonic-like state.
Key Transcriptional (Yamanaka) factors include:
Oct4 (POU5F1)
Sox2
Klf4
c-Myc
Once reprogrammed, iPSCs closely resemble embryonic stem cells in:
Morphology
Gene expression profiles
Epigenetic state
Differentiation potential
However, unlike ESCs, iPSCs are not derived from embryos therefore making them ethically less controversial and more broadly acceptable.
Key properties of iPSCs
There are two fundamental characteristics of iPSCs:
Pluripotency
They can differentiate into derivatives of all three germ layers:
Ectoderm
Mesoderm
Endoderm
Self - Renewal
They can proliferate indefinitely while maintaining an undifferentiated state under appropriate culture conditions.
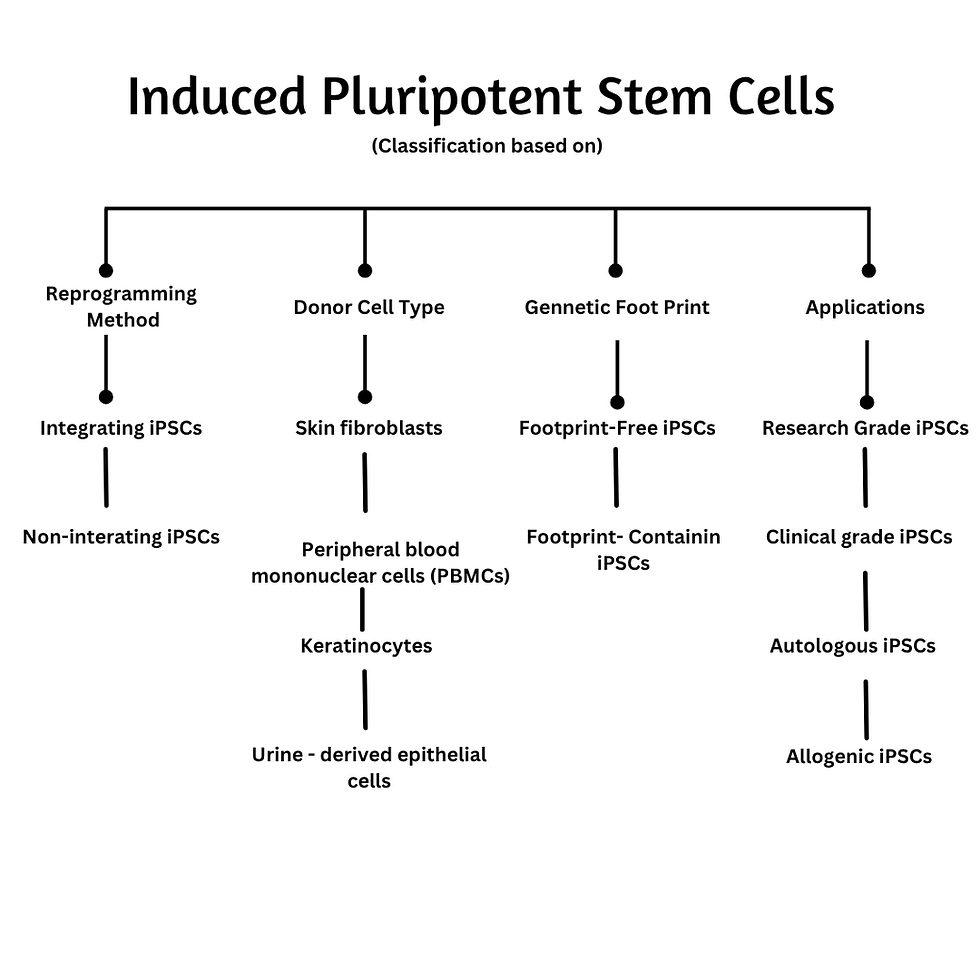
Classification of Induced Pluripotent Stem Cells
iPSCs can be classified based on reprogramming method, donor cell type, genetic footprint and intended application. This classification is critical for understanding their safety, efficiency and translational potential.
Classification based on reprogramming method
a. Integrating iPSCs : These iPSCs are generated using vectors that integrate into the host genome. For example using retroviral vectors or lentiviral vectors show high reprogramming efficiency but also have limitations like risk of insertional mutagenesis and oncogene activation.
b. Non-integrating iPSCs: Reprogramming factors are delivered without permanent genomic integration. For Example using sendai virus, episomal plasmids, mRNA transfection or protein - based delivery show improved genomic safety which widely preferred for clinical applications. However limitations could include lower efficiency with high technical complexity.
Classification based on donor cell type
iPSCs can originate from various somatic sources, each influencing reprogramming efficiency and epigenetic memory.
Common donor cells include:
Skin fibroblasts
Peripheral blood mononuclear cells (PBMCs)
Keratinocytes
Urine-derived epithelial cells
Cells closer to a progenitor state often reprogram more efficiently and retain less residual epigenetic memory.
Classification based on genetic footprint
a. Footprint-Free iPSCs: These cells contain no residual exogenous genetic material after reprogramming. Therefore they reduce the risk of genomic instability and increase their potential to be used in therapeutics.
b. Footprint -Containing iPSCs: Cell contain residual sequences from vectors or transgenes. Therefore they can be used in basic research or for disease modeling.
Classification based on application
a. Research-Grade iPSCs
Used for developmental biology
Disease modeling
Drug screening
b. Clinical-Grade iPSCs
Generated under Good Manufacturing Practice (GMP) conditions
Genetically stable
Stringently characterized
c. Autologous iPSCs
Autologous (patient-specific): Reduced immune rejection
d. Allogeneic iPSCs
Allogeneic: Scalable and cost-effective, but may require immunosuppression
Why iPSCs matter in Brain Research
In neuroscience, iPSCs allow research to:
Model neurodevelopmental disorders in human cells
Study patient-specific neurodegenerative diseases (e.g., Parkinson's, ALS)
Generate neurons, astrocytes and organoids for mechanistic studies
Screen neuroactive compounds in disease-relevant systems
Unlike animal model, iPSC-derived neural cells preserve human-specific genetic and epigenetic features.
Conclusion
Induced pluripotent stem cells have redefined what is possible in stem cell biology. By enabling the generation of patient-specific, pluripotent cells without ethical contovery, iPSCs bridge basic research and clinical translation.
Understanding how iPSCs are classified- by reprogramming strategy, donor cell source, genetic footprint, and application - is essential for evaluating their strengths, limitations, and future potential. As reprogramming technologies improve, iPSCs are poised to play a central role in personalized medicine and regenerative therapies, particularly in the study and treatment of brain disorders.


Comments